Surgical lighting infrastructure is one of the most persistent drains on ASC budgets. Legacy fiber optic systems carry hidden costs that compound across hundreds of annual cases: cable maintenance, thermal risks, sterilization labor, and workflow delays. For distributors selling into this market, understanding these cost drivers is the difference between a transactional sale and a consultative partnership.
This article examines the total cost of ownership for wired vs. cordless illumination systems and quantifies the ROI case for high-volume surgical facilities.
Selling into the sterile processing department is the step most surgical device reps skip. It’s also where deals collapse after a surgeon has already said yes.
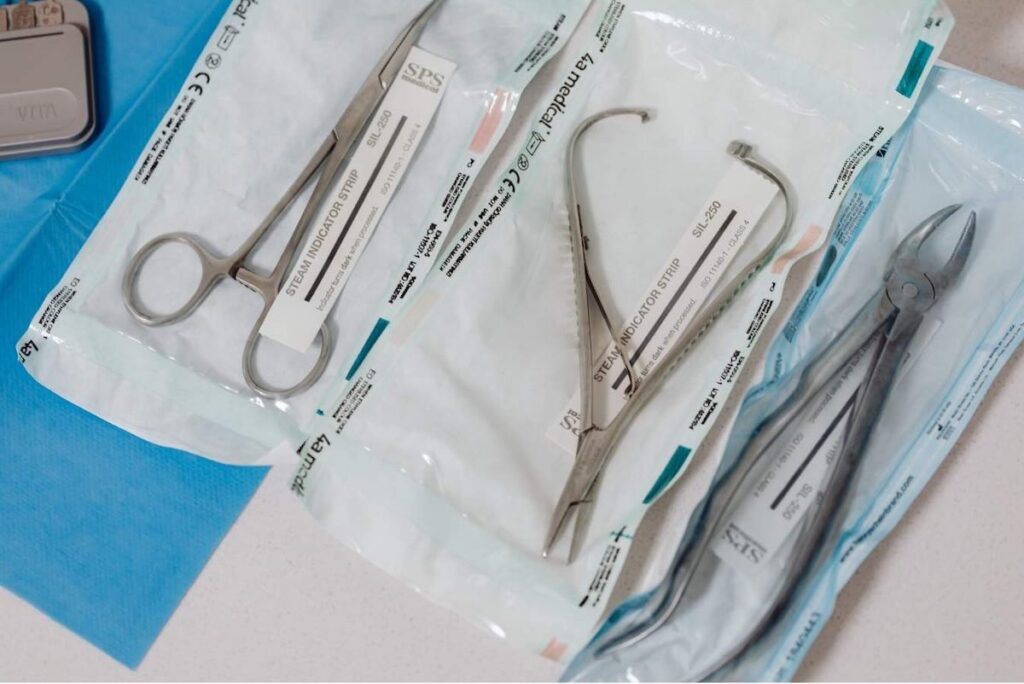
SPD controls what gets approved, reprocessed, and stocked, and it can veto a purchase at any point in the approval chain, including after the OR manager has signed off. Understanding who holds that veto, what SPD evaluates before approving a new instrument, how fiber optic lighting cables typically fail that evaluation, and how to structure the SPD conversation will get you in front of deals that most reps never see. Distributors who know this process don’t just close more deals; they become the rep the facility calls first.
Contents
Who actually controls the device approval process?
SPD controls the final step in device approval, not the surgeon. The buying chain runs from surgeon request to OR manager sign-off to value analysis committee to SPD, in that order. Surgeon preference gets the conversation started, but SPD determines whether a device can actually be integrated into the facility’s reprocessing workflow.
The structural reason SPD gets excluded is simple: reps call on clinicians, not processing technicians. OR relationships are easier to build and more immediately rewarding. But the approval process doesn’t end in the OR. AORN’s 2025 Sterilization Guideline formalized what SPD has always done informally: the department must confirm that a new instrument’s sterilization method is feasible and compatible with existing equipment before the purchase is finalized.
AORN perioperative specialists have documented that instruments are frequently purchased without SPD input, and the consequences show up later as delays, workarounds, and compliance gaps. A 2021 study in BMJ Quality & Safety reviewed 41,799 surgical cases and found that 5% were affected by instrument tray defects, with missing instruments and tray composition among the leading causes.
If you get to SPD before the purchase is finalized rather than after the first reprocessing cycle fails, you close the deal without the reversal. You also position yourself as someone who understands how a hospital actually runs, which matters to purchasing managers who’ve been burned by reps who didn’t. What SPD needs to say yes is where that conversation starts.
What does SPD need to say yes to a new instrument?
SPD needs three things: a sterilization method that works with equipment already in the department, a turnaround time that fits the daily case schedule, and an IFU that technicians can actually follow. That’s the checklist every new instrument goes through.
FDA’s 2015 Reprocessing Guidance requires manufacturers to provide validated instructions covering the full reprocessing cycle. SPD technicians are expected to follow those IFUs as written, so an IFU that specifies equipment the facility doesn’t have, or that runs to multiple pages of steps spread across different components, creates a reprocessing problem before the device sees a single patient. The Joint Commission lists sterilization as its most frequently cited high-risk accreditation standard, which gives facility administrators a compliance reason to take SPD’s objections seriously.
Fiber optic lighting cables tend to fail on all three counts. Ethylene oxide sterilization, which many fiber optic systems require, takes roughly 2.5 hours of processing plus 8–12 hours of aeration per CDC guidance, meaning the cable sits in processing for most of a working day. EtO is also classified as a hazardous air pollutant under EPA regulations and carries OSHA occupational exposure limits that require engineering controls and medical surveillance programs, infrastructure that many ASCs simply don’t have.

Vaporized hydrogen peroxide systems are faster but don’t penetrate long coiled lumens reliably, per CDC. Pre-use visual inspection for fiber breakage is a standard IFU requirement for fiber optic cables, which adds technician time to every cycle. And a 2015 study found that 50% of sterile surgical light handles cultured positive during hip arthroplasty, with Staph epidermidis and Staph aureus the most common isolates.
When you show up with IFU documentation, sterilization validation sheets, and cycle compatibility data, you shift the conversation from evaluation to implementation. That’s a different meeting than the one most reps get.
How do cordless LED handles reduce SPD’s workload?
Cordless LED handles address each of the problems fiber optic cables create for SPD: they process faster, they carry a simpler IFU, and the component that contacts tissue is disposable.
The koplight™ is one example of how this works in practice. The handle carries an IPX7 waterproof rating, which allows it to go through an automated washer without the manual cleaning process that fiber optic cables require. VH2O2 sterilization cycles run 24–74 minutes with no aeration and no hazmat handling, per FDA clearance data and CDC guidance, a turnaround window that fits most facility schedules. For SPD managers juggling tight daily timelines, that difference in cycle time affects how many sets the department needs to keep in inventory.
The single-use blade takes the highest-bioburden component out of reprocessing entirely. The blade contacts tissue, gets discarded after the case, and never enters the SPD cycle. What remains is a waterproof handle with a straightforward IFU covering a single component. That’s easier to train technicians on, easier to document for accreditation purposes, and less likely to produce the assembly errors that delay cases.
Long turnaround times on fiber optic systems also force facilities to overstock inventory to cover daily case volume; a faster reprocessing cycle reduces how many sets the department needs on hand. A study on instrument tray simplification found that reducing component complexity at one surgical center saved 383.5 SPD labor hours and $69,441 in resterilization costs in a single year. For total cost-of-ownership comparisons with a purchasing manager, average SPD reprocessing runs around $58 per tray.
Once you’ve made the case on reprocessing, the question is how to get the right people in the room.
How should distributors approach the SPD conversation?
Request a meeting with the SPD manager directly, not just the OR coordinator. Many reps never do, and that’s a gap worth exploiting.
OR coordinators are the path of least resistance. SPD managers are the ones who flag sterilization incompatibilities, hold up procurement, and, when they’re on your side, become internal advocates who make the VAC conversation considerably easier. A good first meeting with an SPD manager doesn’t require a full product demonstration. It requires showing up with the IFU, knowing which sterilization cycles the product is validated for, and being able to answer the three core questions:
- Can we sterilize it?
- How long?
- How complex?
If you can answer those clearly, you’ve done more than most reps who’ve tried to sell into that facility.
Where an infection preventionist is involved in purchasing, include them from the start. APIC formally directs IPs to collaborate with SPD and value analysis committees on infection prevention, and contamination data tends to carry more weight in those conversations than it does with OR coordinators. The finding that half of sterile surgical light handles cultured positive is exactly the kind of data point worth raising with an IP.
Frame the meeting around the metrics SPD managers actually track. A 2024 study found that 26% of surgical cases had at least one instrument error, with an average delay of 10 minutes per case. A distributor who connects reprocessing simplicity to fewer errors and faster turnaround gets a different meeting than one who leads with clinical features.
Bring the documentation: IFU sheets, sterilization validation data, cycle compatibility specs. For distributors presenting the koplight™, that documentation covers a two-component system (waterproof handle and single-use blade), which is a straightforward reprocessing conversation compared to a multi-component fiber optic setup. Value analysis committees expect the paperwork. SPD managers remember the reps who showed up with it.
Add the koplight™ to your portfolio
The koplight™ is a cordless LED lighted retractor with a waterproof handle and single-use blades. FDA-registered and EU MDR certified. Distributors interested in adding it to their lineup can contact Yasui directly.


